Synthesis Attachment Chemistry
Synthesis Attachment Chemistry.
Building with Biochemistry
Whether performed in-house or as a commercial service; synthesis efficiency and sequence
integrity depends on two major chemical components; attachment chemistry and ancillary reagents.
- The biochemical properties at the base level of synthesis are generally not well known, or they are ineffectively presented.
- This piece provides a technical introduction regarding the chemical components involved in nucleotide synthesis
- The information provided will help serve as a starting point for delving into technical biochemistry and its relation to DNA and RNA synthesis.
Attachment Chemistry & Ancillary Reagents
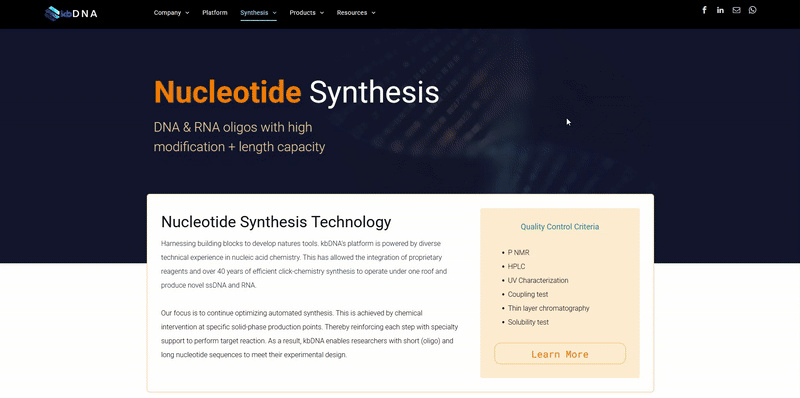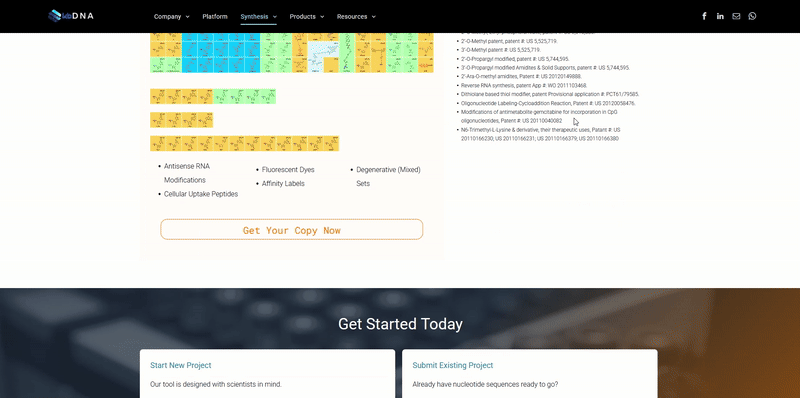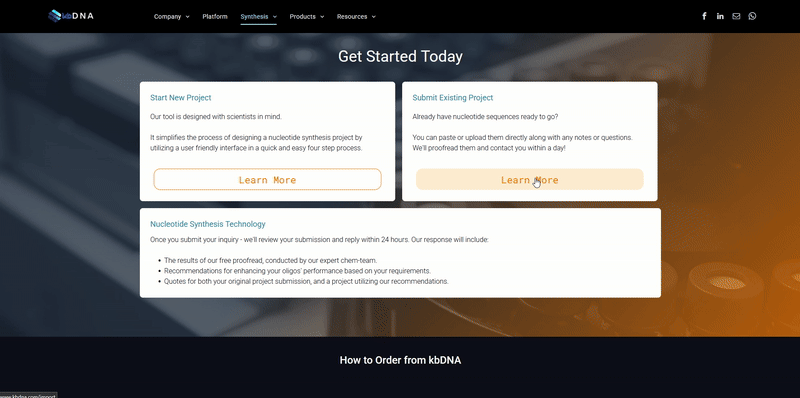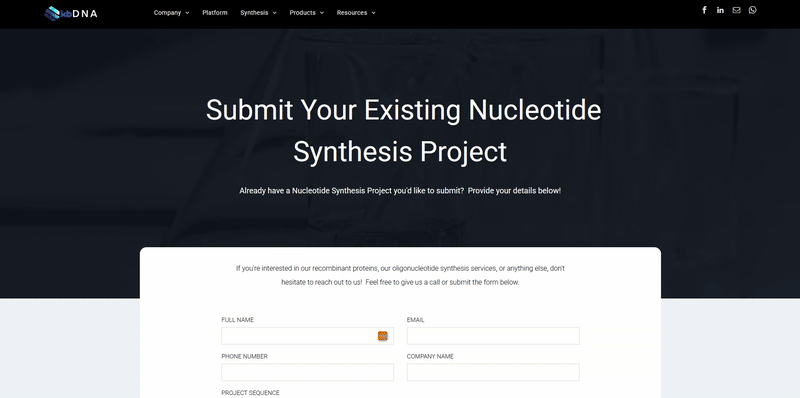
Attachment Chemistry involves two classes of reagents; amidites and supports. These are the primary building blocks of nucleotide synthesis. Their properties and features help construct sequences to meet specific objectives. Their level of quality is one of the tell tale signs of a company’s synthesis expertise.
To support the mechanisms of attachment, ancillary reagents are produced for specific applications. These reagents play an essential role in synthesis optimization. They are formulated to ensure performance within their respective stage and can help direct the synthesis process.
Amidites & Supports
A simpler way to understand nucleotide synthesis is to regard it as the attachment of one amidite to the next via a phosphate backbone. This results in a chain of amidites or nucleotides. Amidites are nucleoside structures that are either carrying a base at the 1' position or modifications labelled on its ribose ring. They are often referred to as phosphoroamidites due to the presence of a phosphate group (synthetic nucleotides) and offer extensive chemical and/or structural modification possibilities.
Supports are much easier to understand since their function is in the name. Supports are protective material that attach to the amidite and follow the reagent along the synthesis process until it is time for removal. The relationship between amidites and supports makes it easy to understand why they are referred to as the building blocks of nucleotide synthesis. However, their varying features present changes in biochemical properties and often require adjustments in handling. It is very important to understand the requirements of every amidite and its modifications to build the right sequence for its application.
It is important to have a reasonable understanding of the intended reagent building blocks for a synthesis project due to their lack of experimental cooperation. Adjustments in protocol concentration, dilutions, temperature and time are often required to achieve proper coupling or yield of differing reagents to individual standard. As an example; kbDNA uses a selection of building blocks that are strictly within their realm of competence. Doing so allows for quality synthesis and the confidence to offer technical support and recommendations for optimization. The selection is a bit more strict than others, however it does offer a great visual of the different patterns that help signify expertise in a particular area of service.
The selection includes:
- Quenchers
- Etheno DNA Amidites and Supports
- Phosphate Generating Reagents
- Biotin Amidites and Supports
- Psoralen Amidites and Supports
- Fluorescein Amidites and Supports
- Etheno RNA Amidites and Supports
- TAMRA and ROX
- Amino Modifiers
- Cholesterol Amidites and Supports
- DNP-TEG Amidites and Supports
- Dabcyl Amidites and Supports
- Branching Amidites & 3'-Spacer Supports
- Spacer Amidites
- 3'-Carboxyl Linker Amidites
- Thiol Modifier Amidites and Supports
- Lipophilic Amidite
Ancillary Reagents
In order to aid attachment chemistry, ancillary reagents are formulated to enhance or guide the synthesis process at target stages. By chemical tweaking and solvent mixing--the resulting reagent mixes and solutions contribute to capping efficiency, positive oxidation reduction, safe deblocking, and more. Contrary to building block reagents, these ancillary chemicals are usually rather standard in their quality. It is difficult to come across competitive differences. However, the useful knowledge required with ancillary chemicals is less about quality and more about qualifications. There are varying mixes out there, and quality tends to come down to matching the right solution with the situation. That is why we recommend organizing your internal lab synthesis chemicals relative to their value and unique chemical properties.
The following is how we stack the ancillary reagents in our chemical arsenal to ensure the qualifying option is applied in each synthesis project:
Capping-Stage Solutions
- CAP A (Acetic Anhydride/Pyridine/THF)
- CAP A (N-Methyl imidazole; 20% in Acetonitrile)
- CAP B (10% N-Methylimidazole in THF)
- CAP B (16% N-Methylimidazole in THF)
- Cap B1 (Acetic Anhydride; 40% (v:v) in Acetonitrile)
- Cap B2 (Symmetrical Collidine; 60% (v:v) in Acetonitrile)
Oxidation-Stage Solutions
- Oxidation Solution (0.02M Iodine/Pyridine/H2O/THF)
- Oxidation Solution (0.05 M Iodine in Pyridine(90%) and Water(10%))
- Oxidation Solution (0.1M Iodine/Pyridine/H2O/THF)
Deblocking-Stage Solutions
- Deblock (3% DCA in Toluene)
- DMT Removal Reagent (3% Dichloroacetic Acid/Dichloromethane)
- DMT Removal Reagent (3% Trichloroacetic Acid/Dichloromethane)
In summary; the process of nucleotide synthesis is heavily dependent on the mechanisms of attachment chemistry. Attachment chemistry involves different classes of reagents, primarily building blocks and ancillary reagents. Building block amidites represent the synthetic form of a nucleotide or modified compound via the reinforcement of a support. Both of these building blocks are promoted and directed throughout each stage of the synthesis cycle by ancillary reagents.
This should help provide a better understanding of the technical components that drive attachment chemistry in nucleotide synthesis. We look forward to building off this note with more technical insight regarding the different mechanisms and method of attachment chemistry. This includes click chemistry, classes of modifications, application protocols, and much more.